5551
Overview
ADAM17 is a metalloprotease that regulates shedding of membrane-bound proteins, including IL-6, TNF-α, and others. ADAM17 is implicated in inflammatory diseases, fibrosis, cancer, and autoimmune conditions, making it a compelling target.
We developed an inhibitor based on ADAM17’s natural pro-domain (TPD), which selectively suppresses ADAM17 activity.
Applications
- Treatment of autoimmune, inflammatory and fibrotic diseases, including inflammatory bowel disease (IBD), rheumatoid arthritis (RA), psoriasis, multiple sclerosis (MS), systemic sclerosis, and lupus
- Cancer therapy through localized inhibition of ADAM17
- Combination with immune checkpoint inhibitors or anti-TNFs
Differentiation
- Highly selective compared to small molecules
- Smaller than an antibody, enhancing tissue penetration
- Anti-inflammatory effects in mouse models of colitis, arthritis & pancreatitis
- Anti-fibrotic in a kidney fibrosis mouse model
- Reduced tumor growth in a lung cancer mouse model
Development Stage
- Inhibitor synthesized and structurally validated
- Showed in vivo benefit in mouse models of colitis, arthritis, kidney fibrosis, pancreatitis, and lung cancer
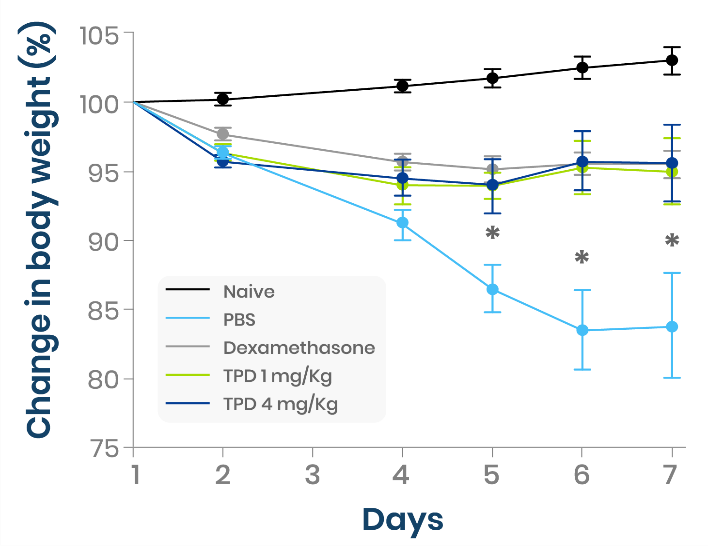
The pro-domain based ADAM17 inhibitor (TPD) prevented weight loss in colitis mice, comparable to dexamethasone
References
Wong, E. et al, Sci Rep., 2016
Eirini Kefaloyianni et al, JCI Insight. 2016
Mohamed I. Saad et al, PNAS, 2022
Contact for more information


