Cancer-associated cachexia (CAC) is a late-stage metabolic syndrome characterized by massive weight loss that occurs in 80% of cancer patients and is responsible for 20% of cancer deaths. It does not respond to dietary changes, and there is currently no effective biomarker or treatment for CAC. Prof. Erez and her team discovered a novel therapy for CAC and created an algorithm that employs regular blood tests to predict the chances of CAC occurrence.
CAC is characterized by the loss of fat tissue followed by skeletal muscle tissue and represents a significant cause of morbidity and mortality in cancer patients, mainly those with in pancreatic adenocarcinoma (PDAC). CAC is associated with immune and central neural system perturbations, resulting in aggravated toxicity complications following cancer therapy. The currently available treatments mainly include nutritional support. However, CAC does not respond to dietary changes. Thus, there is no effective treatment for CAC and no biomarkers for identifying patients at risk for CAC.
Prof. Erez and her team showed that re-expression of a key metabolic regulator known as HNF4α alleviates CAC in a PDAC mouse model. They also developed an algorithm that predicts PDAC patients’ likelihood of developing CAC based on routine blood test results.1
The Weizmann team found that during early carcinogenesis of non-liver cancers (namely breast and pancreatic cancers), innate immune cells infiltrate the liver, causing the depletion of a master metabolic regulator, HNF4α, leading to systemic metabolic changes that promote cancer proliferation and worse outcome. Re-expressing HNF4α with Adeno Associated Virus (AAV8) preserves liver metabolism in a PDAC mouse model and alleviates CAC (Figure 1). Similarly, using HNF4α mRNA encapsulated in lipid nanoparticles (LNPs) successfully alleviated CAC in a PDAC mouse model. The team also developed an algorithm that provides a biochemical liver score that predicts the likelihood of PDAC patients developing CAC based on routine blood test results independent of the disease stage at diagnosis.
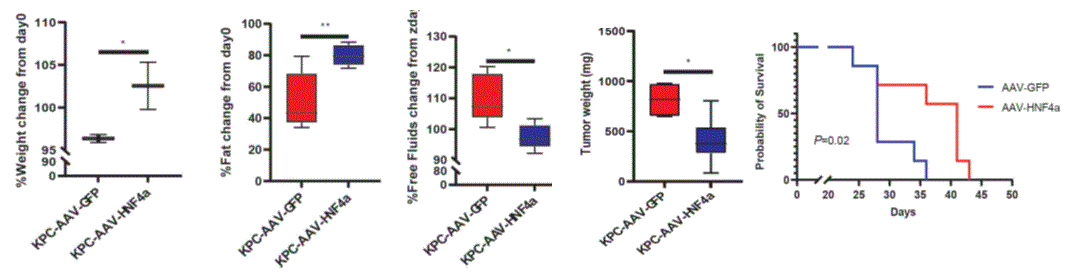
Figure 1 – AAV8-HNF4α virus injected to pancreatic cancer mouse model (KPC mice) significantly alleviated CAC, restricted PC tumor growth and improved survival.
- Treatment for CAC
- Early diagnosis of CAC-inclined patients
The team is currently optimizing the LNP-encapsulated mRNA in a mouse PC model.
Goldman O, Adler LN, Hajaj E, et al. Early infiltration of innate immune cells to the liver depletes HNF4a and promotes extra-hepatic carcinogenesis. Cancer Discov. Published online March 27, 2023:CD-22-1062. doi:10.1158/2159-8290.CD-22-1062 https://doi.org/10.1158/2159-8290.CD-22-1062
Pending

