Cancer, and especially cancer metastasis, is a leading cause of death worldwide. Research in recent years highlighted the importance of the extracellular matrix (ECM) surrounding the tumor in cancer progression metastasis formation. Therefore, many attempts currently address the unmet need to target enzymes that remodel the ECM to treat cancer. The current technology is a monoclonal antibody that specifically inhibits QSOX1, an enzyme that remodels the ECM and is up-regulated in several cancers. The antibody effectively inhibited tumor progression and metastasis formation in several mouse models, especially when combined with chemotherapies. The antibody potentially offers a generalized treatment for several cancer types as a single agent or in combination with standard of care chemotherapies.
Cancer is a leading cause of death worldwide, accounting for almost 10 million deaths in 2020. The primary cause of death from cancer is metastasis,1 demonstrating that despite the ongoing developments in cancer research, there is still an unmet need for therapeutic agents with significant efficacy against solid tumors and metastases formation. The significant role of the tumor microenvironment in tumor development has become evident in the past few years. The ECM is a substantial component of the niche surrounding cells and affects tumorigenesis and metastasis. Therefore there is a need for ECM-modulating agents for treating cancer. Laminin is an important protein of the ECM, as it plays a central role in cell adhesion and migration. Interestingly, laminin is up-regulated in invasive breast carcinomas and was shown to contribute to tumor progression.
Consequently, drugs that target ECM composition by inhibiting laminin incorporation hold great potential as a broad-spectrum treatment for various cancers and metastasis. QSOX1 is an enzyme that is essential for the incorporation of the protein scaffold laminin into the ECM. Cells lacking the QSOX1 enzyme show reduced cell adherence and perturbed migration.
The group of Prof. Deborah Fass developed a monoclonal antibody (mAb) that specifically inhibits extracellular QSOX1 enzyme’s activity.
Quiescin sulfhydryl oxidase (QSOX) catalyzes the insertion of disulfide bonds into unfolded, reduced proteins. QSOX1 expression is up-regulated in the stroma of aggressive breast carcinomas and in a variety of adenocarcinomas, including breast, lung, pancreas, and prostate.2 The group of Prof. Deborah Fass previously demonstrated that knockdown of QSOX1 reduced ECM laminin content, resulting in decreased cell adhesion and reduced cell migration of cancer cells.3 The group further developed a monoclonal antibody (mAb) that specifically inhibits QSOX1 activity by sterically blocking the enzyme's active site. They showed that the antibody affected the ECM produced by cancer-associated stromal fibroblasts, resulting in decreased tumor growth and metastasis in murine cancer models. Importantly, the antibody had an added benefit when provided together with chemotherapy (Figure 1)2.
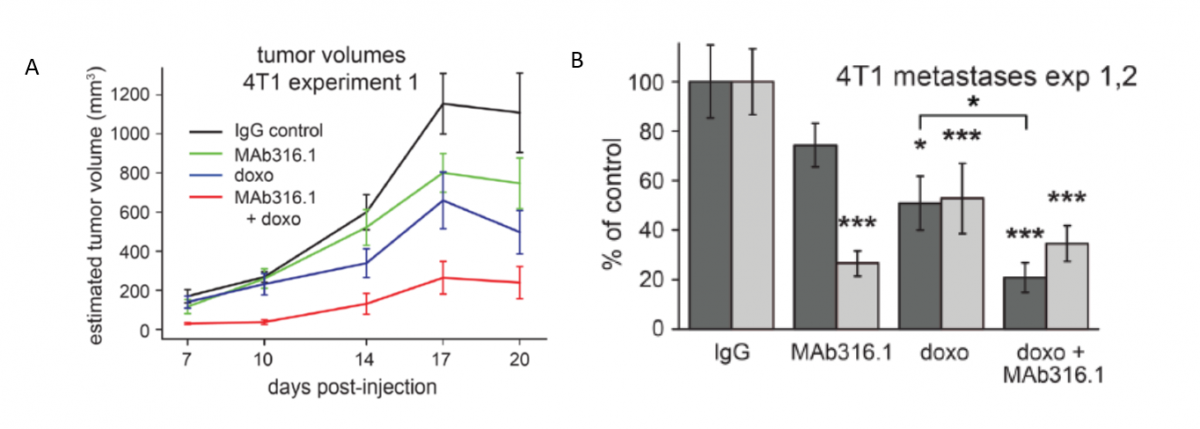
Figure 1. Syngeneic triple-negative breast cancer (TNBC) model treated with QSOX1 inhibitory monoclonal antibody and chemotherapy. (A) Mice bearing 4T1 tumors were treated with control IgG, MAb316.1 alone (30 mg/kg), doxorubicin (8 mg/kg), or a combination of doxorubicin and MAb316.1. Reported tumor volumes were measured externally on the indicated days and averaged for each treatment group. (B) Number of lung metastases in treatment groups from two experiments with the 4T1 model. (* < 0.05; *** < 0.001).
- Therapeutic agent for treating various cancers and inhibiting metastasis
- Can possibly be combined with existing treatments for an enhanced effect
- A possible use for treating laminin-associated diseases
- The mAb only targets the extracellular fraction of QSOX1 without affecting its intracellular activities, which have important physiological roles (in contrast to a small molecule inhibitor).
Prof. Fass and her team have characterized the effect of QSOX1 inhibition on the adhesion and migration of cells. They developed an antibody specifically for QSOX1 (with cross-reactivity to human and mouse QSOX1), demonstrated its effectiveness in QSOX1 inhibition, and showed it decreases tumor growth and metastasis in murine cancer models (syngeneic breast cancer model, syngeneic melanoma model, and xenograft model of human breast cancer). In addition, the group has developed a partially humanized version of the mAb, and is actively working on the development of a fully human version.
Feldman T, Grossman-Haham I, Elkis Y, et al. Inhibition of fibroblast secreted QSOX1 perturbs extracellular matrix in the tumor microenvironment and decreases tumor growth and metastasis in murine cancer models. Oncotarget. 2020;11(4):386-398. doi:10.18632/oncotarget.27438
Ilani T, Alon A, Grossman I, et al. A Secreted Disulfide Catalyst Controls Extracellular Matrix Composition and Function. Science. 2013;341(6141):74-76. https://doi.org/10.1126/science.1238279

