We disclose a novel approach to bypass dose-limiting toxicity of 4-1BB agonists, the major limiting factor for the clinical development of 4-1BB mAbs for cancer immunotherapy: mutating 4-1BB ligand to attenuate its potency and fusing it to an antibody targeting defined immune cells which contributes to efficacy but not toxicity.
This novel bispecific 4-1BBL fusion molecule enhances the efficacy of 4-1BB by exploiting its synergistic effect with the targeting antibody while sparing liver toxicity.
Strategy involves the development of a PD-1+ cell-selective 4-1BB agonist, able to enhance anti-tumor activity while avoiding liver toxicity.
- Optimal therapeutic window of 4-1BB therapy for cancer treatment
- Synergistic effect by combining two therapeutic pathways (PD-1 blockade and 4-1BB activation)
- Platform approach: attenuated 4-1BBL can be combined with the antibody of choice
- Selectivity of 4-1BB agonism to defined immune cell population
- Increased therapeutic window compared to trademark 4-1BB agonists
- Validated in preclinical models, including mice with MC38 colorectal tumors, mice with B16-F10 melanoma and MCA-205 fibro sarcoma
- Fully human fusion antibodies tested in humanized mice and are available for clinical development
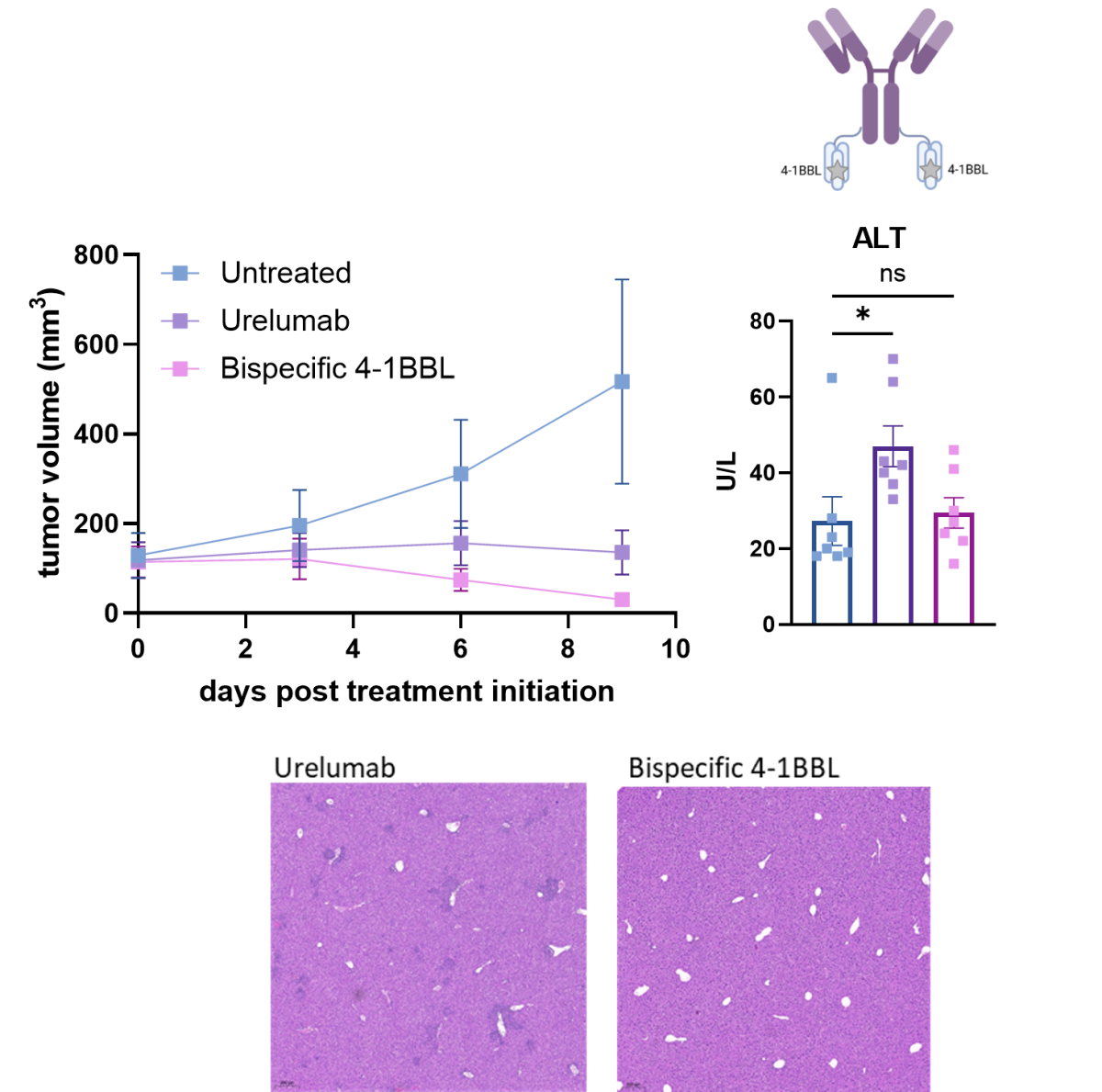
Bi-specific aPD-1-4-1BBL fusion protein show potent anti-tumor activity similar to Urelumab in humanized 4-1BB mice bearing MC-38 colorectal tumors,
with no signs of liver toxicity.


