UV curing of polymers plays a critical role in technologies such as dentistry, advanced manufacturing (e.g., 3D printing), optical media production, and biotechnology. Traditional methods rely on UV irradiation of photoinitiators, but the resulting thermosets are often brittle and limited to thin, transparent formulations. This technology introduces semiconducting nanoparticles (e.g., WS₂) that generate radicals under light to initiate efficient polymerization and cross-linking, resulting in improved mechanical properties and broader applicability. Given their benign characteristics, they can be used also for reinforcing various biocompatible polymers used in medical technologies.
- Reinforcement of radiation curing of acrylates
- Cation-induced polymerization of epoxies for adhesives
- Matrices for fiber composites and 3D printing resins
- Enhances the mechanical properties (elasticity, yield strength, and elongation) of thermosetting polymer films
- Increased film fracture toughness
- Improved film peeling resistance, strength, and adhesion
- Accelerated photocuring process
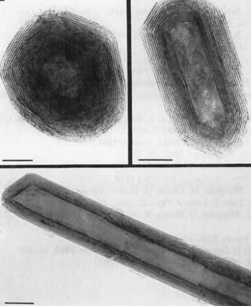
Electron micrographs of polyhedral nanocrystals of WS2
The technology has been demonstrated with dispersed tungsten disulfide fullerene-like nanoparticles (WS2-IF) in acrylate and epoxy resins photocuring, showing covalent bond formation between the nanopaticles and the matrix, resulting in improved adhesion and reduced viscosity.

Dr. Vered Pardo Yissar

