Technologies that target cells or modify their behavior are of great interest to improve diagnostic, therapeutic, and imaging platforms. However, methods for modifying cells' surfaces with synthetic agents are still limited by the complexity of synthesis procedures and restricted dynamic control of the newly acquired features. The proposed technology engineers artificial, self-assembling oligodeoxynucleotide-conjugated functional motifs, which selectively and reversibly bind cell surfaces. The anchored artificial receptor system can respond to external cues and regulate proteins. Conjugates can be easily tailored, local concentrations readily controlled, and functional motifs quickly exchanged, enabling extensive programmability and high applicable potential.
Cell surface proteins are key mediators of cell responses to changing environments. They are naturally regulated by dynamic structure, expression levels, and composition changes. Synthetic agents bearing unique functionalities can also be used to modify and control cell surface proteins. This is commonly achieved by designing biomimetics that bind natural proteins or by incorporating synthetic anchors to the cell surface, which are then chemically or enzymatically modified to acquire the desired functionality. Potential applications include detecting surface protein interactions and turning cells into living scaffolds or platforms for drug discovery. The functional entity can also be introduced via non-covalent interactions, such as protein-DNA interactions or Watson-Crick base pairing. However, versatile and straightforward techniques for synthesis and binding control are still lacking.
Prof. Margulies and his team developed a method to decorate live cells with synthetic oligodeoxynucleotide (ODN)-based surface anchors that provide cells with new properties. These agents are stimuli-responsive protein binders or functional motifs, and external signals can reversibly control their structure and dynamics.
This technology engineers dynamic artificial receptor systems that integrate principles of both ODN-small molecule conjugates and functional synthetic agents. A hexahistidine tag (His-tag) is fused to cell surface-expressed protein, enabling its interaction with artificial nitrilotriacetic acid (NTA)-nickel (II) complex-tagged ODNs linked to a receptor or functional motif. The local concentration of synthetic receptors/functional motifs can be controlled by adding suitable chelators, which disrupt the His tag-mediated bacteria-biomimetic interaction.
Dynamic functionalization of cell surfaces is achieved by decorating cells with a His-tagged ODN (ODN-1), followed by exposure to a complementary ODN (ODN-2) bearing a functionality motif of interest. Functionality is then exchanged by sequentially incubating cells with a strand complementary to the short overhang region of ODN-2 (ODN-3), followed by the introduction of an ODN-2 bearing an alternative functional motif.
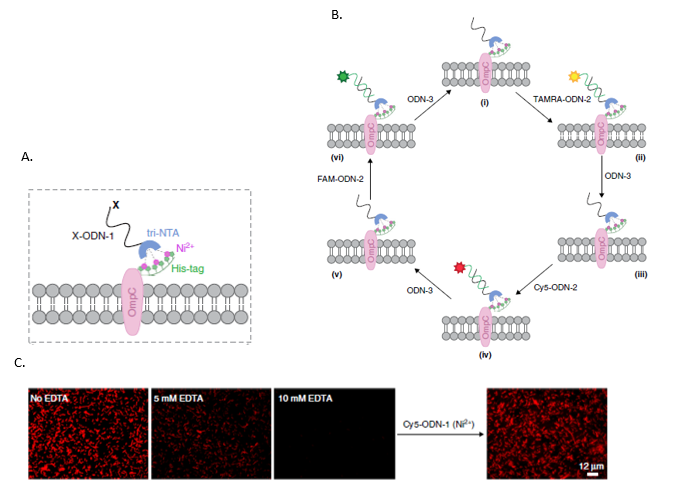
A. A schematic representation of a His-tag fused to OmpC , a protein expressed on the outer membrane of E. coli cells, linked to a X-conjugated ODN-1 through a tri-NTA group. The X-conjugated ODN-1 can be attached to any motif linked to the complementary strand, as shown in figure B. B. Dynamic alteration of the functionality demonstrated by sequentially subjecting cells to His-ODN-1, a fluorescently labeled ODN-2 (e.g., TAMRA-ODN-2), and then ODN-3 that removes the fluorescent ODN-2. The cells can then be relabeled by subjecting them to ODN-2 bearing a different fluorescent group (e.g., Cy5-ODN-2 or FAM-ODN-2). C. OmpC conjugated to a His-tag and incubated with Cy5-conjugated ODN-1. Fluorescence imaging attested to the selective labeling of bacterial cells by Cy5-ODN-1, which did not affect cell viability. Introduction of varying concentrations of EDTA, a potent nickel chelator, effectively controlled the degree of synthetic receptor coverage. From: Lahav-Mankovski et al., Nature Commun. 2020,11 :1299
Advantages
- Simple synthesis
- High versatility
- Allows co-detection of different factors
- Selective and reversible binding
- Precise control of orientation, distances, and valency of binding units
- Dynamic, real-time control of cell surface functionalization
- Control of local concentrations of receptors/functional motifs
Applications
- High-throughput drug screening/direct binding studies
- Cell imaging methods
- Cell-based therapeutics/diagnostics
- Living materials and devices
This platform's extensive variability and programmability were demonstrated (published in: Lahav-Mankovski et al., Nature Commun. 2020,11 :1299). The team is currently working on tailoring this technology to cancer cell-targeting and drug screening platforms. The researcher will be willing to cooperate with other companies to integrate this technology in other specific applications.
The Global Drug Screening Market is constantly on the lookout for new technologies and developments. In 2019 it was valued at USD 7.09 billion and is projected to reach USD 14.51 billion by 2027, growing at a CAGR of 10.1% from 2020 to 2027, according to a recent report. At the same time, the increasing adoption of automated platforms and innovative products is streamlining diagnostic lab activities and enabling the early diagnosis of various disorders. The growing incidence of infectious and chronic diseases has significantly enlarged the clinical diagnostics, which is expected to grow at a CAGR of 5.2 between 2019 to 2024. In particular, the global cancer diagnostics market is expected to reach USD 249.6 billion by 2026.

Dr. Vered Pardo Yissar

