5596
Overview
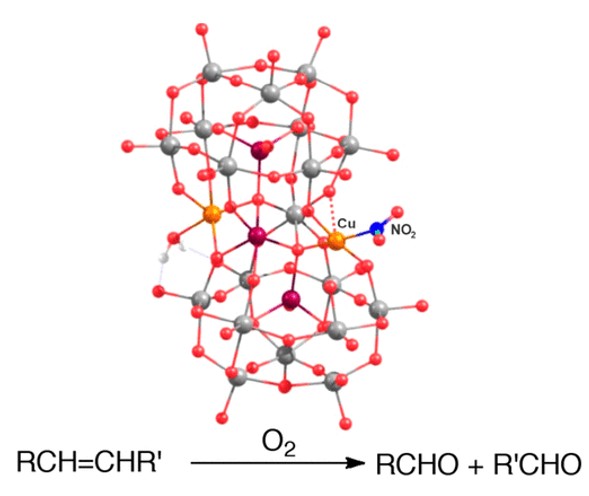
A novel green chemistry approach for producing ketones and aldehydes through an oxidative cleavage reaction. The process utilizes polyoxometalate (POM) catalysts to achieve aerobic cleavage of carbon-carbon double bonds in alkene.
Applications
- Industrial organic chemistry processes for oxidation reactions.
- Renewable feedstock processing, such as unsaturated fatty acid derivatives.
- Stand-alone catalysts for laboratory or individual use.
Differentiation
- Scalable
- Use renewable resources as feedstock
- Environmentally Friendly
- Easy catalyst regeneration
Development Stage
A proof of concept was demonstrated on a lab scale.
Patent Status:
USA Granted: 9,815,757
Contact for more information

Dr. Vered Pardo Yissar
Senior Director of Business Development, Exact Sciences

