5533
Overview
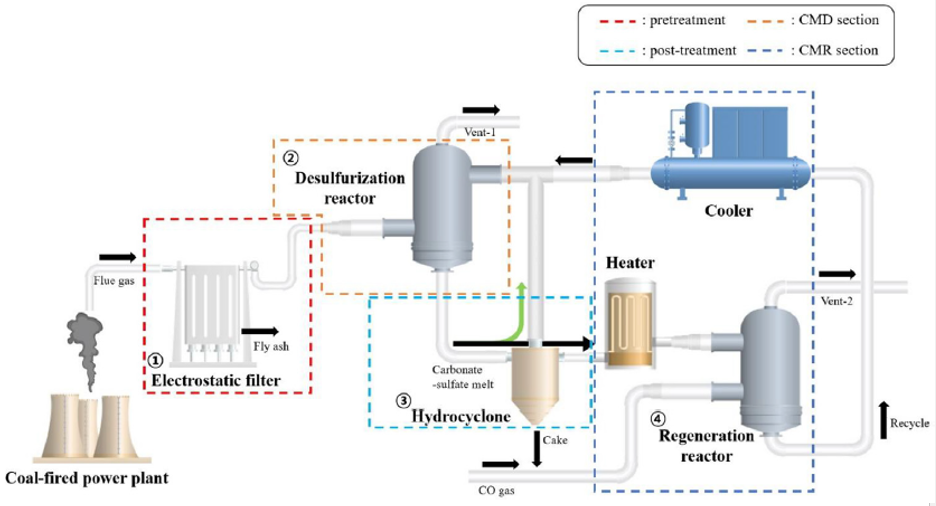
Current sulfur dioxide (SO₂) removal technologies are outdated, costly, and environmentally hazardous. This novel system offers a high-efficiency, low-waste solution for purifying industrial flue gases by converting SO₂ into reusable sulfur compounds. Operating via a carbonate eutectic melt at moderate temperatures, it enables selective recycling of SO₂ without producing slurry waste, thus aligning with future environmental standards and creating potential for sulfur valorization.
Applications
- Power plants (coal-fired)
- Cement factories
- Metallurgical facilities
- Industrial flue gas treatment systems
Differentiation
- High SO₂ removal efficiency (residual SO₂ in ppm range)
- No generation of hazardous slurry waste
- Operates at standard industrial temperatures (480–550°C)
- Compact system suitable for retrofitting (e.g., ~tens of m³ for 1GW plant)
- Enables revenue generation via production of sulfur/sulfuric acid
Development Stage
Proof-of-concept prototype validated in lab and successfully tested in a coal-burning power plant.
Patent Status:
USA Granted: 8,852,540 USA Granted: 10,625,204
Contact for more information

Dr. Vered Pardo Yissar
Senior Director of Business Development, Exact Sciences

