A rapid, label-free biosensor for detecting specific conformation of His-tag labeled proteins, accessible for use in both academic- and commercial-R&D was invented at the Weizmann Institute of Science. This fluorescent biosensor detects and labels His-tagged proteins in complex environments, such as living cells, while offering high sensitivity, speed, and cost-effectiveness.
Fluorescent biosensing has become a valuable tool in analytical research, while at the same time the poly-histidine tag (His-tag) has become the most commonly used tag for protein labeling. However, commercial products for labeling and detecting conformational changes in His-labeled proteins suffer from three main limitations: First is their inability to identify specific changes in the protein conformation. Second, even if such changes are detected, labeling of His-tag proteins is usually very laborious. Third, commercial probes tend to be large and consequently can interfere with the proteins function. Therefore, there is a need for a small, direct, and non-interfering fluorescent probe for detection of conformational changes.
Prof. Margulies and his team have developed a first of its kind fluorescent molecule that provides detection of changes to the surface of His-labeled proteins. Subsequently, labeling proteins only if they are in a specific conformation. The biosensor exhibits high affinity, excellent signal-to-noise ratio and can be easily applied with no fixation or tedious protocols.
Using the technology, many proteins and specific conformations can now be labeled and studied for the first time (proteins that were already successfully labeled using the biosensor: Calmodulin, B-cell lymphoma 2, G-protein).
The biosensor presented here is comprised from three main molecular-components: (I) the selective binder (binds to a specific tag, e.g. His-tag); (II) the nonselective binder (binds to the protein surface, as long it is in a specific conformation); and (III) a solvatochromic fluorophore.
The selective binder (I) ensures the sensor will only bind to a tagged protein (in this case, His-tagged protein), regardless of its conformational state. Once the protein folds into the specific conformation, the nonselective binder (II) detects and binds to the modified surface configuration (pre-designed to detect the specific conformation). Now that the local environment of the attached-biosensor and its fluorophore (III) has changed, the fluorophore subsequently emits a distinct and measurable signal.
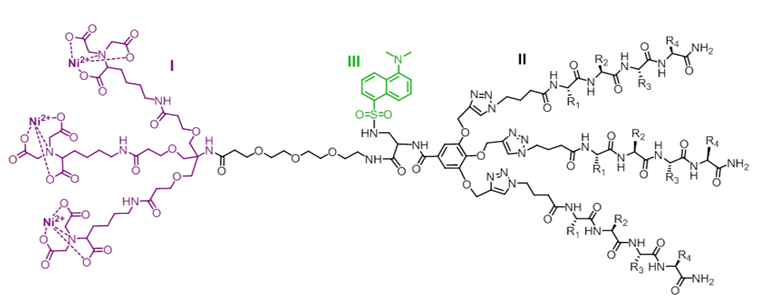
An example of a protein-surface sensor consisted of a tri-Ni2+-NTA complex (I), a tripodal peptide receptor (II), and a dansyl group (III), which serve as a His-tag binder, a protein-surface receptor, and a solvatochromic probe, respectively.
Advantages
- Directly, rapidly, and easily applicable.
- Usable in complex biological environments – live cells or biological media.
- Demonstrates high affinity and superior signal-to-noise ratio.
- Small in size - unlikely to interfere with the labeled protein function.
Applications
- Fluorescent detection of specific conformations of His-tag labeled proteins.
- Direct labeling in both live cell imaging and biochemical assays.
- Tracking protein-protein interactions in vivo.
- Localizing proteins and measuring their steady-state concentration in vivo.
- High throughput screening of inhibitors of protein-protein interactions in living cells
The methodology of creating fluorescent molecular sensor responding to changes occurring on the surfaces of specific proteins was successfully demonstrated (Published in: Chem. Eur. J. 2015, 21,15981). The basis of this technology was later demonstrated in living cells expressing his-tagged proteins showing effective and reversible operation. Other project expanding the applications of this invention are currently in progress. Two filed patent applications cover this invention.

Dr. Vered Pardo Yissar

