Acute liver failure (ALF) is a rapid deterioration of liver function, resulting in a high mortality rate in the absence of liver transplantation. ALF treatment remains highly limited and mostly supportive due to a lack of a comprehensive understanding of the molecular base of ALF. The group of Profs. Eran Elinav and Ido Amit used high-resolution single-cell analysis methods to unravel the entire molecular mechanism that drives ALF progression. They show that ALF results from activation of the MYC signaling pathway in specific hepatic cells in response to gut microbiome signals. Based on their findings, inhibition of MYC, TLR, or MAPK pathways or microbiome modulation by antibiotics could attenuate ALF.
Acute liver failure (ALF) is a devastating, rapidly progressing disease that results in death in 80% of cases unless an emergency liver transplant is performed. ALF may result from multiple infectious, immune, metabolic, and neoplastic diseases. In the developed world, its leading cause is a substantial overdose of acetaminophen, also known as paracetamol. Although ALF is extensively studied, a comprehensive understanding of the molecular basis of ALF is lacking. Notably, the gastrointestinal tract drains directly into the liver and exposes the liver to substances produced in the gut and by its microbes. Thus, the gut microbiome directly affects hepatic physiology and pathologies. Therefore, there is an unmet need to fully characterize the molecular mechanism that drives ALF by the liver tissue itself as well as the gut microbiome for developing novel treatments for ALF.
The groups of Profs. Eran Elinav and Ido Amit conducted a comprehensive high-resolution single-cell gene expression analysis of liver cells from mice ALF models. Their study revealed that ALF is driven by the MYC signaling pathway that is activated in specific subsets of hepatic cells in response to signals from the gut microbiome.
The groups of Profs. Eran Elinav and Ido Amit profiled the gene expression of 45,000 individual mouse liver cells, ultimately generating high-resolution liver cell atlas under healthy and ALF conditions. The scientists identified that in the course of ALF progression, abnormally activated cells arose among three cell subsets: stellate, endothelial, and Kupffer cells. These activated cells secreted cytokines that attracted immune cells from outside the liver, which then contributed to its damage. All the above-mentioned activated cells shared an expression pattern controlled by the transcription factor MYC. Inhibition of MYC signaling resulted in reduced haptic damage and extended mice survival (Figure 1). To evaluate the potential role of the microbiome on ALF development, the teams depleted the microbiome of the mice by administering wide-spectrum antibiotics, and by examining germ-free mice. The depletion of the microbiome attenuated MYC activation and alleviated symptoms of ALF (Figure 2). Furthermore, the researchers showed that distinct molecules generated by the microbiome (MAMPs) and damaged liver cells (DAMPs) accumulated in the liver and induced TLR signaling, which activated the MAPK pathway, resulting in the MYC signaling activation that contributed to liver damage. Indeed, inhibitors of MAPK signaling by specific IRAK4, TAK1, and p38 inhibitors resulted in a significant reduction of ALF associated damage (monocyte infiltration, Serum aspartate transaminase (AST), and alanine transaminase (ALT) activity).
Finally, the researchers analyzed liver samples from patients with ALF and compared them with samples from healthy liver donors. Only the liver samples from ALF patients were characterized by robust MYC activation, similar to the observation in mice. These results raise the possibility that blocking the MYC program by drugs, coupled with microbiome modulation, may prove to be a potential treatment for acute liver failure1.
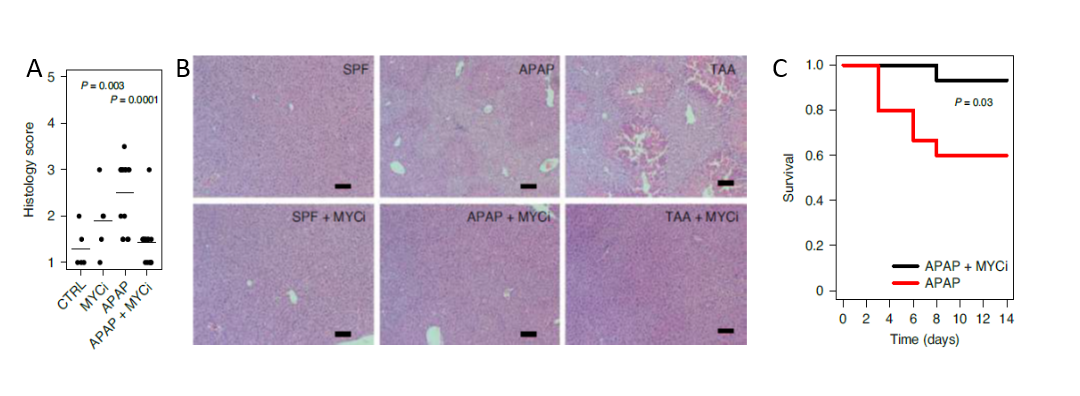
Figure 1 – Inhibition of MYC signaling resulted in reduced haptic damage and extended mice survival A. Histology scores of H&E-stained liver sections from TAA liver failure model in the presence or absence of MYC inhibitor. (n = 10). B. Representative H&E-stained liver sections. Scale bars, 100 μm
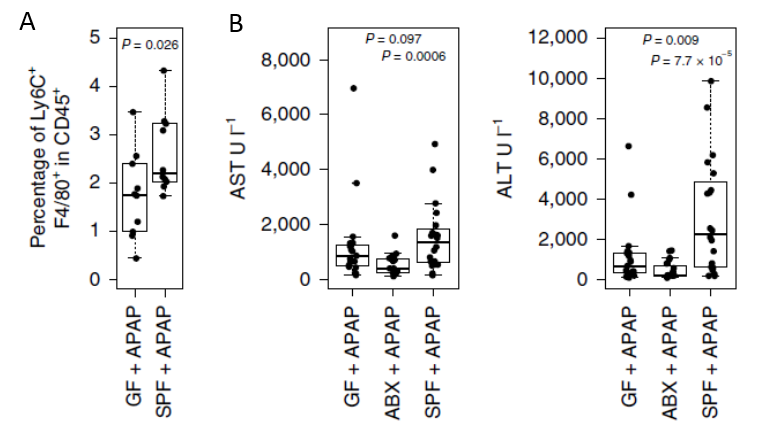
Figure 2 - The microbiome modulates response to acute insult via MYC. A. FACS analysis of the percentage of Ly6C-positive monocytes within all immune cells in germ-free (GF) and control (SPF) mice(n = 10). B. Activity of Serum aspartate transaminase (AST), and alanine transaminase (ALT) in mouse serum from ALF model in GF, ABX, and SPF mice.
- Treatment for ALF
The groups characterized the entire molecular mechanism that drives ALF in two mice models and demonstrated its relevancy in human liver samples.
References:
Kolodziejczyk AA, Federici S, Zmora N, et al. Acute liver failure is regulated by MYC- and microbiome-dependent programs. Nat Med. 2020;26(12):1899-191 doi:10.1038/s41591-020-1102-2


