CoLDR (Covalent Ligand-Directed Release) chemistry enables site-specific covalent labeling and activation of endogenous proteins using small-molecule covalent ligands. Upon binding to a target cysteine, the directing ligand is released and a functional cargo, such as a drug, fluorophore, degrader, or affinity tag, is installed while maintaining the protein’s native activity. This versatile platform provides precise chemical control for targeted drug activation, imaging, and protein manipulation in live cells without genetic modification.
- Targeted prodrug activation in cancer and inflammatory diseases
- Drug and probe delivery with selective activation in target-expressing cells
- High-throughput screening using CoLDR-based luminescent or fluorescent probes
- Proximity-inducing therapeutics, including PROTACs, PHICs, and molecular glues
- Combinatorial or synthetic-lethal drug release strategies
- Target-activated release: cargo released only upon covalent binding
- Activity preserved: enables functional labeling without inhibiting proteins
- Tunable and modular: adaptable chemistry and broad range of cargos
- Broad scope: demonstrated on >10 proteins including BTK, KRAS-G12C, EGFR and more
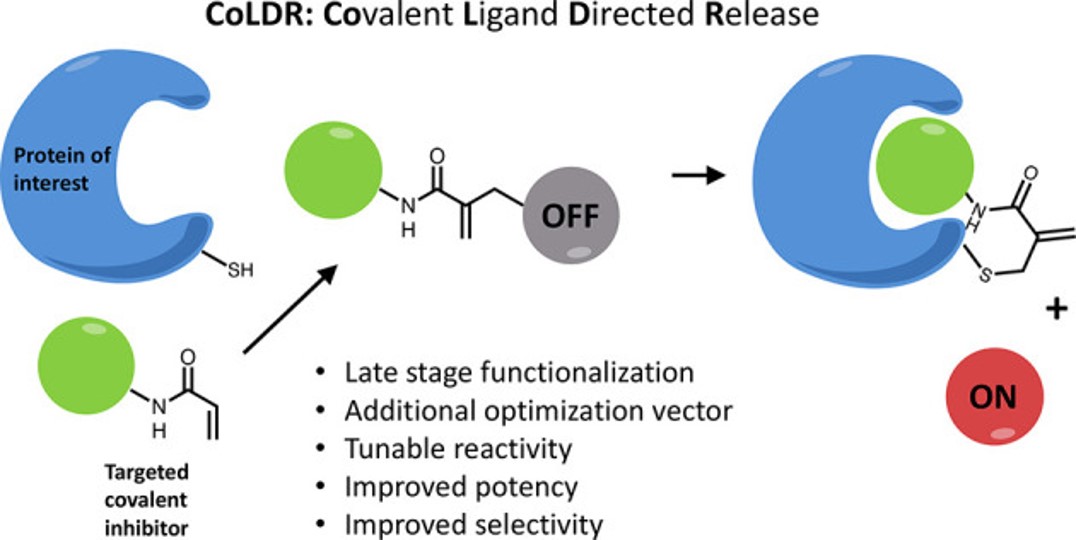
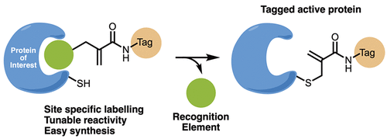
Proof-of-concept shown in vitro, in cells, and in vivo.
CoLDR probes selectively label target proteins with fluorescent, alkyne, or luminescent tags, enabling visualization and tracking while preserving activity.


